Poster
Deepening understanding of tumor budding in colorectal cancer for effective immunotherapy
Posted on:
Colorectal cancer (CRC) is one of the most prevalent cancers and causes of cancer-related deaths. The histologic type of cancer, the disease’s stage, the tumor-node-metastasis (TNM) staging system, and the status of the circumferential resection margin (CRM) are typically used to determine the best course of treatment. However, they only give a partial picture because many patients with projected early-stage disease have systemic and lymph node micrometastases, which may lead to disease recurrence1. These findings highlight the shortcomings of current techniques for accurately stratifying patients with CRC and the urgent need to find biomarkers that can provide clinicians with insights into prognosis and therapy choices.
What is tumor budding?
Tumor budding, defined as the existence of a single tumor cell or clusters of up to 4 tumor cells, is an emerging prognostic biomarker in CRC2,3. It has been linked to epithelial-mesenchymal transition (EMT) in the tumor microenvironment (TME) of CRC (Figure 1) and poor disease-specific survival rate4.
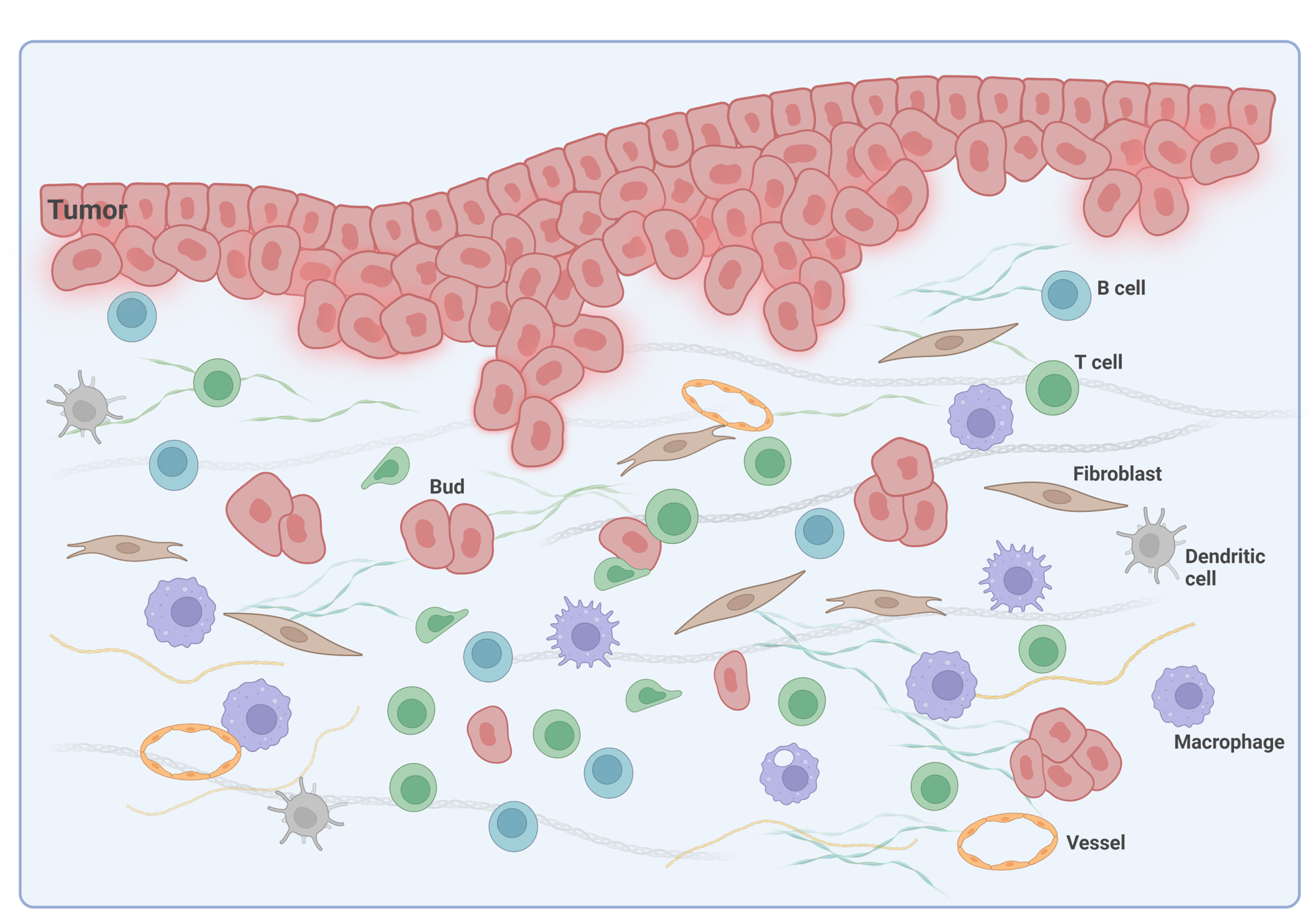
In pT1 colon cancer patients, tumor budding categories are utilized to determine if to conduct radical surgery after endoscopic resection. In stage II colon cancer, tumor budding is an indicator considered for the guidance of adjuvant treatment decisions5, which is a form of treatment used in conjunction with primary therapy in order to increase the effectiveness of both. In stage II CRC, it was also demonstrated that the spatial interaction between T lymphocytes and tumor budding holds significance as individuals with more lymphocytes around tumor buds show a better prognosis6. The recent post-hoc analysis of the International Duration Evaluation of Adjuvant Chemotherapy (IDEA) France phase III study demonstrated that tumor budding also offers additional and clinically relevant prognostic information in patients with stage III colon cancer5.
Related Articles
Cytokines, spatial activation and tumor infiltration: the future of immunotherapy
Posted on 13 Dec 2023
Read Post