Poster
Cytokines, spatial activation and tumor infiltration: the future of immunotherapy
Posted on:
Cancer immunotherapy is a constantly advancing research area, playing a vital role in combating cancer. A central aspect of this field is the study of cytokines, essential signaling molecules that coordinate immune responses against malignancies. However, utilizing cytokines specifically and effectively in cancer therapy is complex and multifaceted. This is where spatial biology comes into play, offering insights into cytokines signaling within the tumor microenvironment (TME). This advanced perspective enhances our comprehension of cytokine-mediated immune responses and refines the strategies for their therapeutic application. This blog thoroughly examines cytokine’s crucial role in cancer immunotherapy. It investigates how advancements in spatial biology support the development of more effective and targeted therapeutics by revealing the intricate interactions and spatial dynamics of cells within tumors.
The role of cytokines in immunotherapy
With the development of immunotherapy, there is a rising emphasis on using innate host defense mechanisms to combat cancer. Among them, cytokines, small signalling proteins secreted by cells, play an essential role in mediating cell communication, localization and interaction. They are produced by various cell types, with helper T cells (Th) and macrophages being the primary sources1. Cytokines actively engage immune effector and stromal cells in the tumor environment, thereby improving the ability of cytotoxic effector cells to recognize and target the tumor. This unique property has led to intensified research efforts to characterize cytokines and utilize their extensive signaling networks for the design of novel cancer therapies2.
Despite the FDA‘s approvals of Interleukin-2 (IL-2) and Interferon-alpha (IFN–ɑ) for cancer treatment, the clinical success of cytokines has been limited3,4. This is primarily due to their restricted therapeutic applications and the occurrence of dose-limiting toxicities5. For example, high doses of IFN–α can lead to severe flu-like side effects characterized by fatigue, fever, headaches, muscle pain, or gastrointestinal issues6. Therefore, a significant contrast exists between the body’s natural, precise use of cytokines and the broader, less targeted approach typically seen in external cytokine therapies. Internally, cytokines are deployed in specific and tightly controlled time, dose, and place, addressing targeted areas as needed. Conversely, external therapies often involve a systemic distribution, which lacks the refined specificity of the body’s innate mechanisms. In addition, cytokines can influence various cell types, and this broad impact can create complexities in developing targeted and, thus, effective therapies.
Recent advancements in protein engineering have emerged as pivotal solutions to these challenges. By fine-tuning the aspects of cytokine therapy — specifically the timing, location, specificity, and duration of cytokine activity — protein engineering is bridging the gap. It enables systemically administered cytokines to resemble more the body’s natural cytokine biology. This approach can potentially enhance the efficacy and safety of cytokine-based treatments, offering a more sophisticated and targeted strategy that aligns with the body’s biological processes, while limiting off-target and side effects.
Advancing cytokine-based therapies through spatial insights
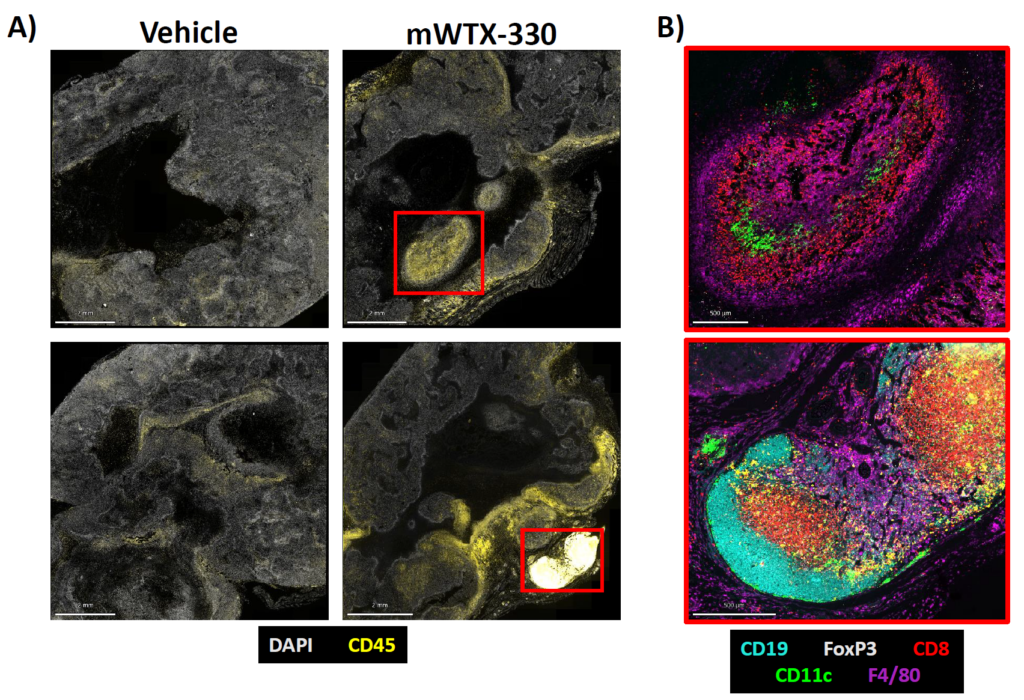
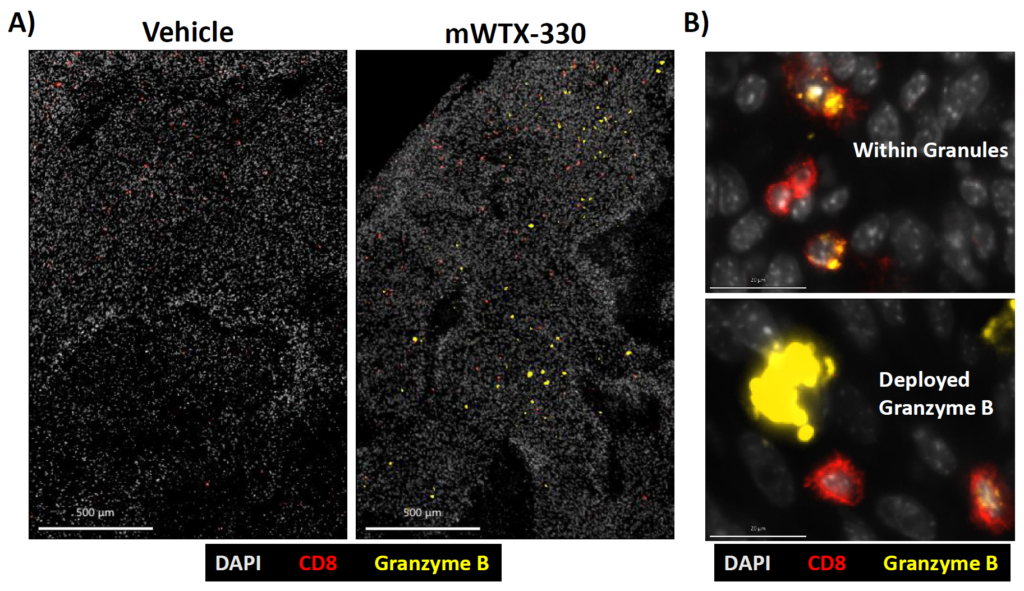
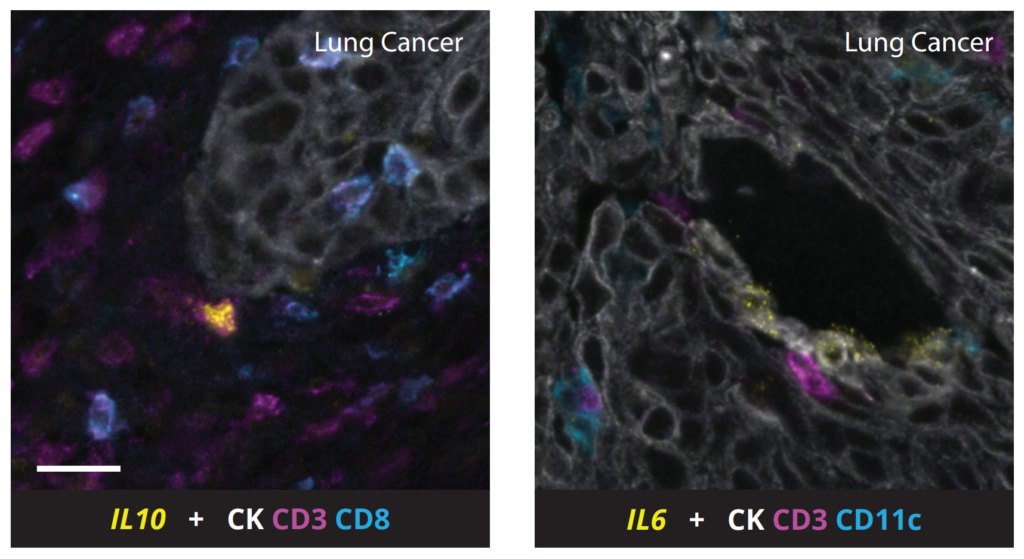
Cancer immunotherapy relies heavily on understanding the tumor microenvironment, where cancer, immune, stroma cells, and other biological and chemical components interact. Multiplex immunofluorescence (mIF) provides a window into these interactions by:
- Characterizing cell populations: it can reveal the presence, abundance, and organization of both immune and stromal cells within the tumor, which is critical for understanding tumor biology and the immune response.
- Analyzing cell activation: it allows scientists to assess the activity state of each of the cell populations through biomarker expression and spatial organization and extract insights on the tumor immunogenicity.
- Highlighting tumor heterogeneity and clinical response: by combining the presence, organization, and activity state of cells composing the TME, it helps understand the tumor heterogeneity and correlate it with the patient’s clinical benefit from cancer therapies.
- Revealing spatial biomarkers associated with therapy response: using mIF to analyze tumor biopsies before and after therapeutic intervention can help identify or validate candidate predictive biomarkers.