Poster
Immunosuppressive brain tumor microenvironment: a barrier to overcome
Posted on:
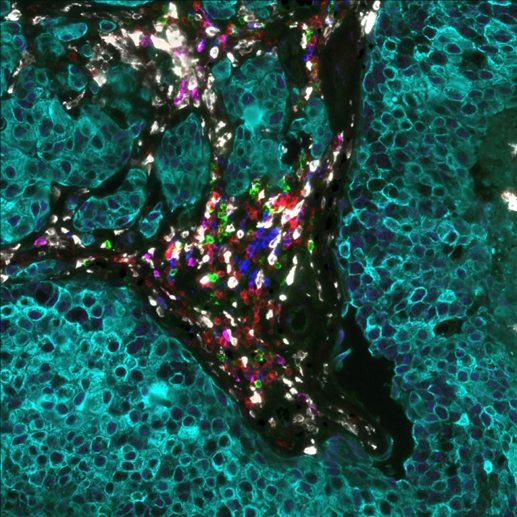
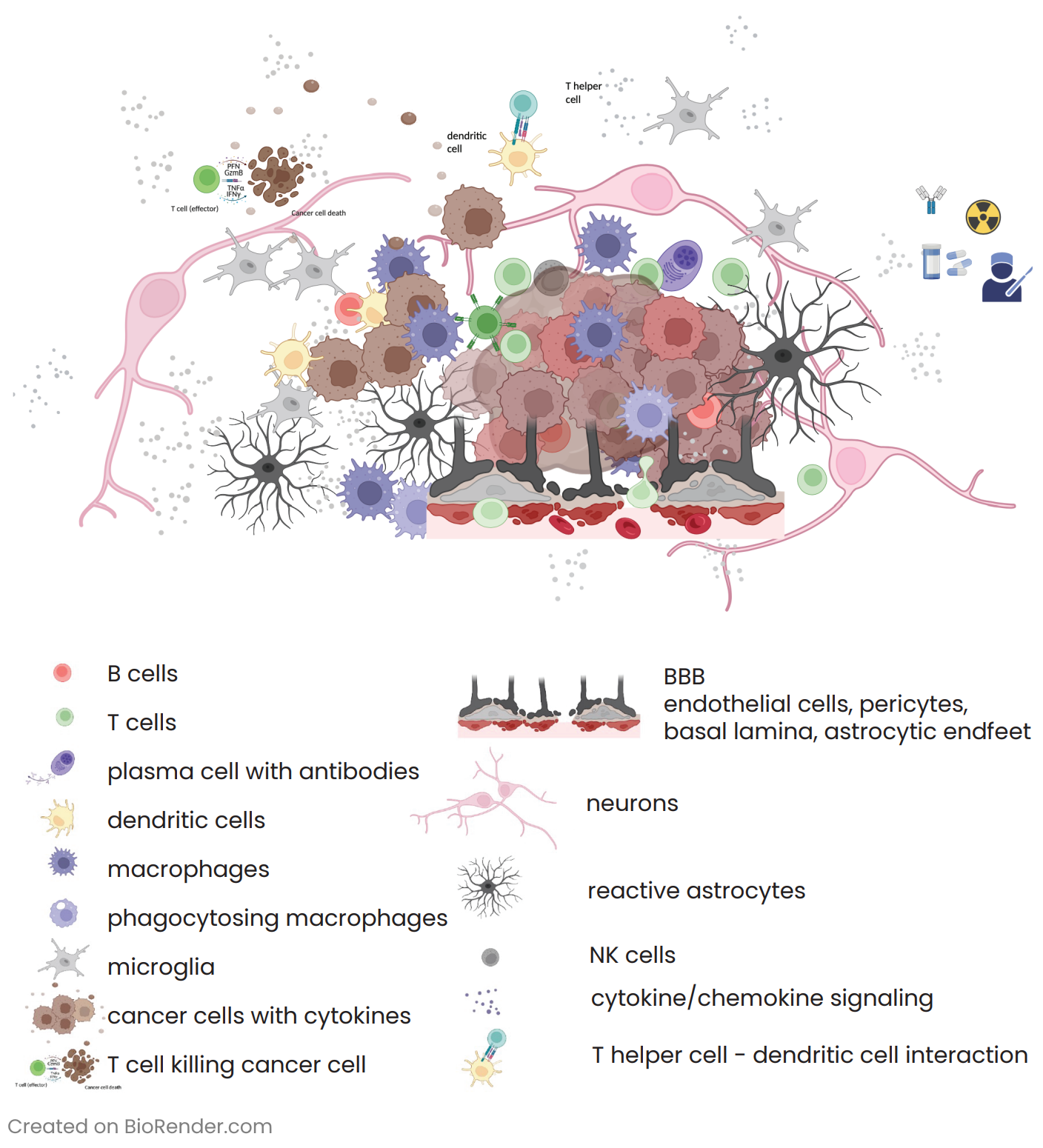
Glioblastoma (GB) and brain metastases (BM) represent the prevalent malignant tumors affecting the central nervous system in adults. For BM, despite the availability of advanced diagnostic techniques and optimal multidisciplinary treatment approaches, which involve surgical resection and radiotherapy (RT)1, the unfortunate reality is that almost all patients eventually face tumor progression. Within the complex realm of brain tumors, an intriguing aspect that researchers have uncovered is the presence of an immunosuppressive tumor microenvironment (TME)2 (Figure 1). The TME creates a unique environment that hampers the immune system’s ability to respond effectively against the tumor. By studying the spatial distribution via multiplex immunofluorescence (mIF) of immune cells, tumor cells, and molecular markers within the brain TME, researchers can gain valuable insights into the mechanisms behind immunosuppression and identify potential therapeutic targets.
Researchers unveil spatial architecture of TME in brain metastasis using seqIF™
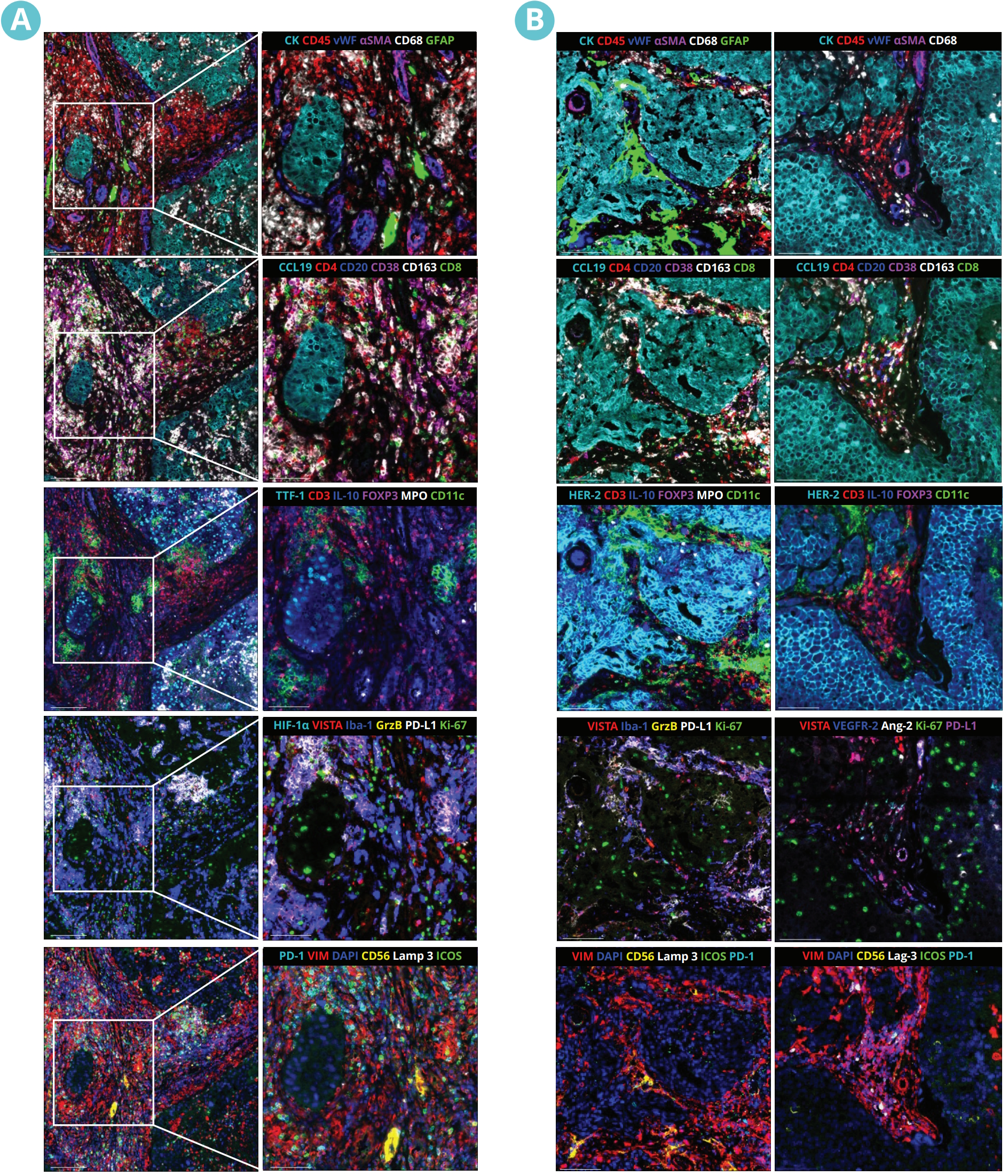
In a recent study presented by the Institute of Neurology (Edinger Institute), University Hospital, Goethe University Frankfurt, Germany, at the AACR 2023 Annual Meeting, researchers investigated the spatial architecture of the TME in lung and breast-to-brain metastasis using sequential immunofluorescence (seqIF™) on COMET™. SeqIF™ offers a novel, straightforward method to detect multiple proteins on a single slide with minimal optimization requirements. The process involves staining a tissue section by incubating it with primary antibodies, then detecting markers using fluorescently labeled secondary antibodies. Then images are acquired via the imaging step, and antibodies are removed through a gentle elution step using an elution buffer. This approach allows additional rounds of staining to visualize new sets of antigens, with the ability to use antibodies from different species within each cycle. By stacking the acquired images and creating overlays, spatial relationship maps can be constructed to examine the spatial distribution of the proteins.
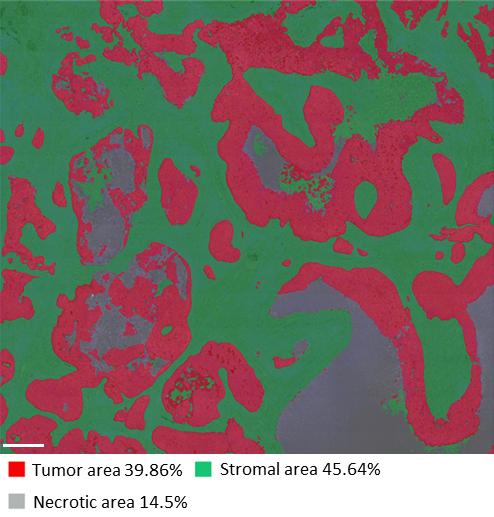
The study was performed using Lunaphore SPYRE™ Immuno-Oncology Core Panel with clinically relevant biomarkers and additional markers targeting myeloid, vascular, and tumor cells, as well as cytokines and immune checkpoints (Figure 2). The high-quality images obtained from the staining panel, encompassing all biomarkers, were analyzed with HALO® Image Analysis platform, developed by Indica Labs, Inc (Figure 3).